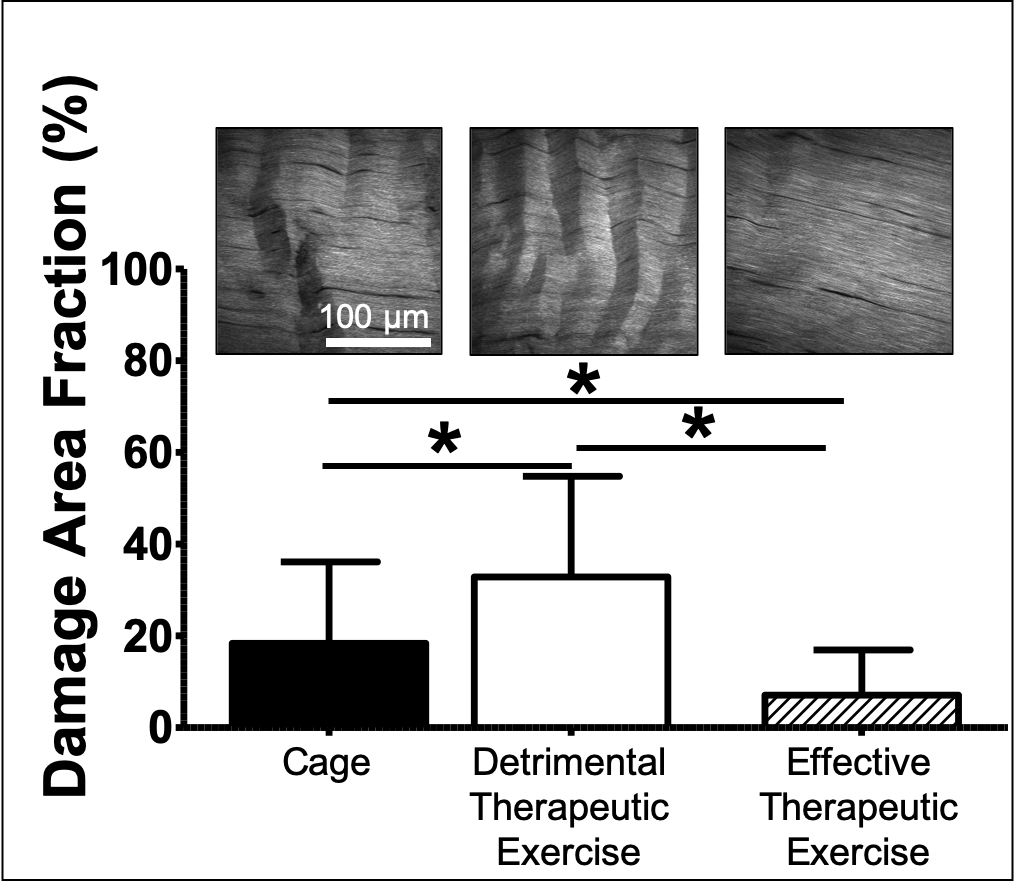
 Onset of fatigue injury leads to prominent apoptotic response (cell death) leading us to suspect that direct inhibition of apoptosis could result in a large available population of cells to repair the induced damage. Our data has shown the contrary, with direct inhibition of apoptosis leading to further degeneration suggesting that forcing cells to remain alive in the context of damaged matrix is detrimental. In contrast, since physiological loading is a powerful modulator of biological mechanisms, we have been evaluating the therapeutic effect of exercise, as well as direct inhibitors of cell death, on the biological environment and effectiveness of matrix remodeling of fatigue damaged tendons. We have found that exercise that is initiated 1-day after fatigue loading leads to further degeneration whereas exercise that is initiated 2-weeks after fatigue leads to repair (see Figure). We have been investigating the biomechanical environment that is necessary for a therapeutic versus detrimental outcome. This data will lead to (1) generation of diagnostics to guide clinicians as to when a tendinopathic patient will effectively repair from physical therapy; and (2) identification of therapeutic outcomes.
Onset of fatigue injury leads to prominent apoptotic response (cell death) leading us to suspect that direct inhibition of apoptosis could result in a large available population of cells to repair the induced damage. Our data has shown the contrary, with direct inhibition of apoptosis leading to further degeneration suggesting that forcing cells to remain alive in the context of damaged matrix is detrimental. In contrast, since physiological loading is a powerful modulator of biological mechanisms, we have been evaluating the therapeutic effect of exercise, as well as direct inhibitors of cell death, on the biological environment and effectiveness of matrix remodeling of fatigue damaged tendons. We have found that exercise that is initiated 1-day after fatigue loading leads to further degeneration whereas exercise that is initiated 2-weeks after fatigue leads to repair (see Figure). We have been investigating the biomechanical environment that is necessary for a therapeutic versus detrimental outcome. This data will lead to (1) generation of diagnostics to guide clinicians as to when a tendinopathic patient will effectively repair from physical therapy; and (2) identification of therapeutic outcomes.
Ongoing projects:
- The role of myofibroblasts in repair of fatigue damaged tendons. We have found that the therapeutic repair outcome from exercise of fatigue damaged tendons is correlated with the increased presence of myofibroblasts as well as several key integrins. Ongoing projects are evaluating the mechanistic role of myofibroblasts in the therapeutic repair process from exercise of damaged tendons.
- The role of glycosaminoglycans (GAGs) in promoting a reparative outcome from subsequent exercise of fatigue damaged tendons. We have previously shown that GAGs are transiently increased after onset of fatigue damage injury. Interestingly, we have also found that exercise that is initiated prior to the increase in GAGs leads to further degeneration of fatigue damaged tendons, but exercise that is initiated during the increase in GAGs leads to repair of the fatigue damage injury. We are evaluating the mechanistic role of GAGs in determining the outcome of subsequent therapeutic exercise.
Relevant publications:
- Bell R, Gendron NR, Anderson M, Flatow EL, Andarawis-Puri N. A potential new role for myofibroblasts in remodeling of sub-rupture fatigue tendon injuries by exercise. Sci Rep. 2018;8(June):8933
- Bell R, Boniello MR, Gendron NR, Flatow EL, Andarawis-Puri N. Delayed exercise promotes remodeling in sub-rupture fatigue damaged tendons. J Orthop Res. 2015;33(6):919-925.